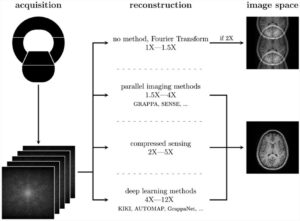
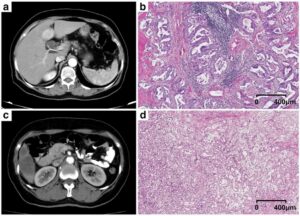
The clinical promise of artificial intelligence (AI) in prostate cancer diagnosis has yet to materialize. Any AI application must reach an appropriate level of maturity and robustness for such developments to be accepted by its intended users. Our position paper, “Development of Artificial Intelligence for Precision Diagnosis of Prostate Cancer Using MRI”, co-authored by experts from ESUR and ESUI, elaborated and compiled the prerequisites for the successful development of AI systems for MRI-based prostate diagnostics.
We have highlighted the following key elements:
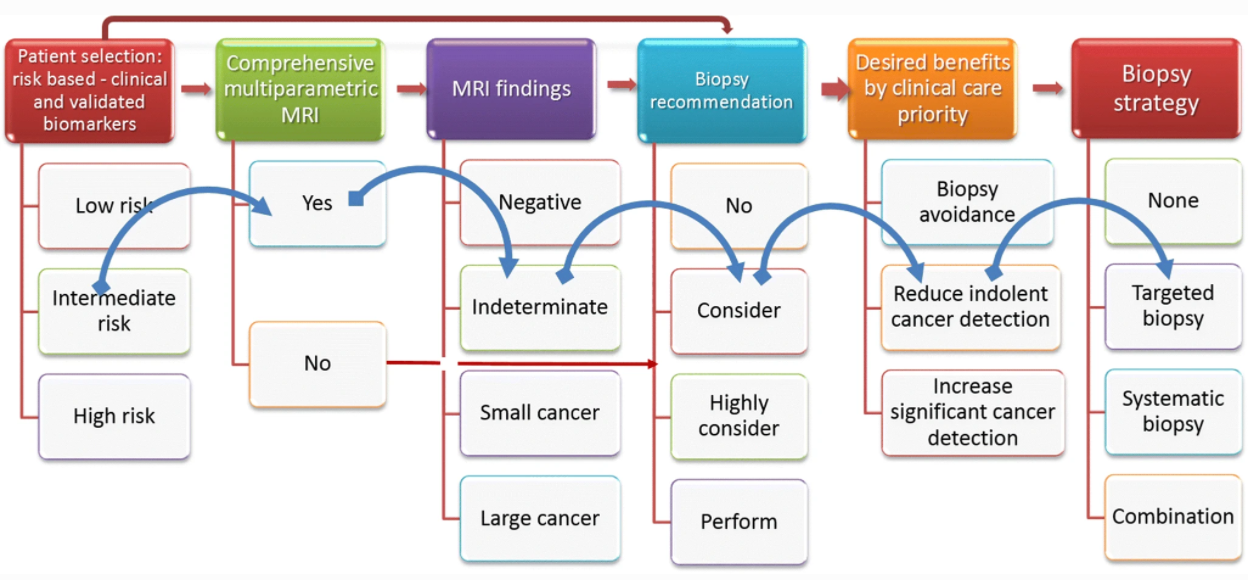
- Systems should ensure that the main benefits of the MRI pathway arising from biopsy avoidance are maintained (consistent & high NPV)
- Systems should increase the consistency of detection of important cancers
- Systems should help improve the discrimination for biopsy of men with indeterminate MRI scans
- Systems should improve radiologists’ productivity by decreasing time-intensive reporting and outlining tasks
- Systems should align with clinical working (stepwise, logically and understandable with actionable outputs)
Finally, a robust implementation that works regardless of MRI vendor or disease prevalence and is responsive to the underlying patient population prevalence and patient preferences is needed.
In our opinion, all of these points are relevant to successfully taking AI in prostate cancer diagnostics into the clinic. We believe that initial work should have focused on developing systems as diagnostic supporting aids for outlining tasks, so they can be integrated into the radiologists’ workflow to support MRI-directed biopsies. The development of AI as a decision support tool will require a larger body of work.
Key points
- AI systems need to ensure that the benefits of biopsy avoidance are delivered with consistent high specificities, at a range of disease prevalence.
- Initial work has focused on developing systems as diagnostic supporting aids for outlining tasks, so they can be integrated into the radiologists’ workflow to support MRI-directed biopsies.
- Decision support tools require a larger body of work including multicentric, multivendor studies where the clinical needs, disease prevalence, patient preferences, and clinical setting are additionally defined.
Authors: Tobias Penzkofer, Anwar R. Padhani, Baris Turkbey, Masoom A. Haider, Henkjan Huisman, Jochen Walz, Georg Salomon, Ivo G. Schoots, Jonathan Richenberg, Geert Villeirs, Valeria Panebianco, Olivier Rouviere, Vibeke Berg Logager & Jelle Barentsz